Periosteal Thickness around the Femoral Neck: a cadaveric study
Manzar H, Antonios TT and Adds PJ
DOI10.4172/2471-8416.100017
Manzar H, Antonios TT and Adds PJ*
Division of Basic Medical Sciences (Anatomy), St. George’s University of London, London, UK
- *Corresponding Author:
- PJ Adds
Institute of Medical and Biomedical Education (Anatomy)
St George’s University of London
Cranmer Terrace
London SW17 0RE, UK
E-mail: padds@sgul.ac.uk
Received date: March 31, 2016; Accepted date: May 02, 2016; Published date: May 05, 2016
Citation: Manzar H, Antonios TT, Adds PJ (2016) Periosteal Thickness around the Femoral Neck: a cadaveric study. J Clin Exp Orthop 2: 17. doi: 10.4172/2471-8416.100017
Copyright: © 2016 Adds PJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: The femoral neck is a common osteoporotic fracture site. In this study we investigate changes in mean periosteal thickness with age, sex, side and circumference along the femoral neck. We hypothesised that: periosteal thickness declines with age, that the periosteum is thicker in males, is thickest inferiorly around the neck, and becomes thicker distally along the neck.
Methods and findings: Sixteen femoral necks from 9 cadavers (mean age 82.78, 5 female, 4 male) were sectioned, decalcified and stained. Periosteal thickness in 4 quadrants around the circumference, and along its length, was measured. The difference in mean periosteal coverage between sexes was found to be insignificant (p=0.2857), and did not appear to decline with age (p=0.7760). No significant changes in periosteal thickness were seen along the length of the neck (p=0.9594). There was, however, a significant difference in coverage around the circumference of the femoral neck (p<0.0001).
Conclusions: The differences in periosteal coverage around the circumference of the femoral neck reported here are open to several theories. There may be potential for pharmacological intervention, gene-related therapy or periosteal transplant to target specifically vulnerable sites.
Keywords
Periosteum; Femoral neck; Osteoporosis; Femoral neck fracture
Introduction
The periosteum plays a major role in bone growth and repair. Bone ossifies by two processes of ossification: membranous and endochondral. In endochondral ossification, the periosteum forms at the periphery of the bone by the perichondrium while in membranous ossification the periosteum forms directly from the mesenchyme [1,2].
Periosteum covers the entirety of the long bones, but is not present on sesamoid bones or intra-articular surfaces of the long bones [3]. In the femoral neck, periosteum is found to be present, and is continuous with the fibrous capsule of the hip joint [4]. The presence of periosteum along the femoral neck accounts for mineralising periosteal tissue, calcifying fibrocartilage and alkaline phosphatase expression along the femoral neck [5-7]. Periosteal circumference has been reported to increase with age, accounting for the growing thickness of the femoral neck in some subjects [8]. However, Shea et al. [9] reported calcification and capsular mineralisation between the femoral head and neck in the elderly, which may account for at least some of the increased femoral neck width seen with age, making it difficult to determine how much of the increase is due to mineralisation and how much is due to periosteal bone formation.
Bone strength and subsequent fracture resistance at the femoral neck is a result of expanding periosteum, which is controlled by osteogenic activity [9]. Power et al. [6] found that alkaline phosphatase was expressed at the femoral neck periosteal surface, which accounts for the mineralisationrelated neck expansion seen with age. Their hypothesis that cases of hip fracture show a lower degree of femoral neck expansion was not proven, since it was found that periosteal bone formation at the femoral neck was a common sign in elderly women, even in the case of hip fractures.
Despite the accepted view that osteoporosis is a consequence of reduced osteogenesis [10], more recent research has found that increased bone resorption is the principle factor in bone fragility [11].
Periosteal resorption has been shown to occur significantly alongside bone growth [3]. Chondrogenesis and periosteal thickness have been shown to decrease significantly with age in rabbits [4]; it is reasonable to suppose that this process also occurs in humans, which would explain the anticipated decrease in periosteal thickness and bone strength seen in old age.
However, absolute bone size is a major determinant of fracture risk and increased bone size comes about by periosteal expansion leading to periosteal thinning. The proliferative nature of the periosteum could leave it open to pharmacological intervention with anabolic or anti-resorptive agents such as those used to increase bone density, and thus reduce the osteoporotic fracture risk.
However, absolute bone size is a major determinant of fracture risk and increased bone size comes about by periosteal expansion leading to periosteal thinning. The proliferative nature of the periosteum could leave it open to pharmacological intervention with anabolic or anti-resorptive agents such as those used to increase bone density, and thus reduce the osteoporotic fracture risk.
Materials and Methods
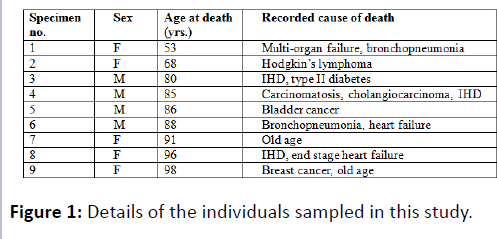
Sixteen femoral necks from 9 cadavers (4 males, 5 females, average age 82.8 years) were retrieved following anatomical examination (2 femoral necks were unusable due to hip replacement surgery) (Figure 1). The cadavers used in this study had been donated for anatomical examination and research under the Human Tissue Act (2004).
The medical practitioners of all the donors were contacted for information on: past history of fracture, osteoporosis risk factors, drugs that increase the risk of osteoporosis, protective drugs against osteoporosis and relevant investigations, including bone fragility scans. There were no significant histories to report.
Sectioning & labelling
The femoral sections were removed from embalmed cadavers in the dissecting room. Following removal of subcutaneous fat and muscle to obtain a clean bone specimen, the femoral neck was removed by making two transverse cuts using an oscillating bone saw.
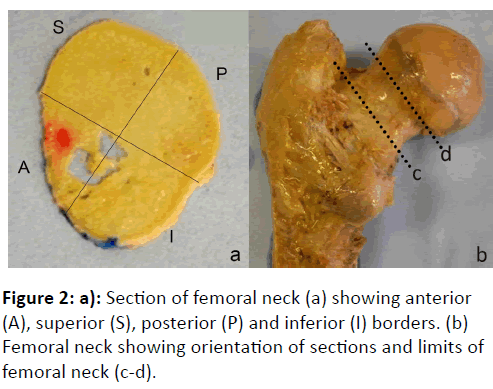
The greater trochanter of the femur was used as a reference point for making the distal cut, parallel with but just proximal to the base of the articular capsule, in line with the highest point of the greater trochanter. The proximal cut was made just below the expansion of the femoral head. The femoral neck specimens were cut into 1 mm sections using a Buehler IsoMet® low speed saw with a diamond wafering blade.
A red histological ink was used to make a dot on the anterior quadrant (on the superior face of the section). The remaining quadrants could then be defined as superior, posterior and inferior, respectively (Figure 2).
Decalcification, Staining & Microscopy
The sections were decalcified in 14% EDTA (ethylenediaminetetraacetic acid, VWR) in PBS (phosphate buffered saline), buffered to pH 7.2 with 0.1 M NH4OH solution. Samples were left to soak in a 1-litre beaker containing the EDTA buffer on a hot plate set at 20°C, and agitated with a magnetic stirrer. The pH of the solution was monitored daily for 3 days and buffered when necessary. After 3 days the sections were removed and stained with Sudan black which highlights nerve fibres in the bone. This stain was found to be effective as the periosteum is dense in nociceptive nerve endings [12].
Stained sections were whole-mounted onto microscope slides with Histomount®, covered with a coverslip and placed in a laboratory oven at 70°C for 24 hours to dry.
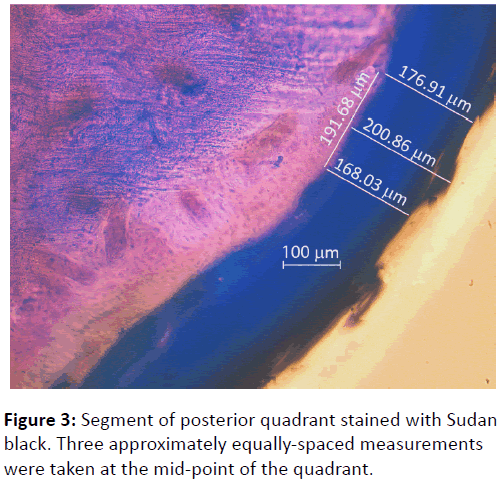
Light microscopy and photography were carried at 10x magnification with a Zeiss Axioplan 2 upright microscope under mild fluorescence to enhance the differentiation between cortical bone and periosteum. Three approximately equally spaced measurements of periosteal thickness were taken from the middle section of each quadrant over a distance of approximately 200 μm (Figure 3). This gave a total of twelve readings from each section. The mean of the three readings from each quadrant was calculated, giving an average measurement of periosteal thickness for the anterior, superior, posterior and inferior quadrants, respectively.
Statistical analysis
All data were statistically analysed using GraphPad Prism 6 software. Mean periosteal thickness was compared to age, sex, side, anatomical border and distance along the femoral neck.
Results
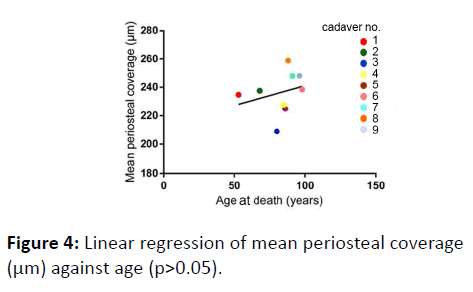
No correlation was found between age and mean periosteal thickness (p=0.7760) (Figure 4).
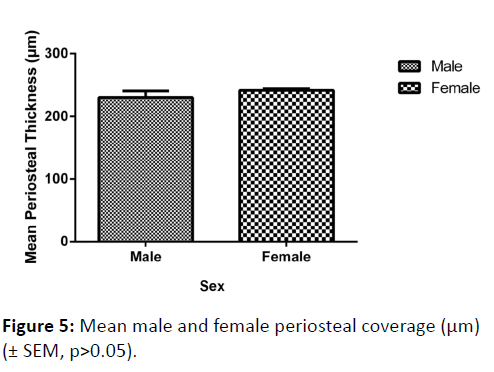
Although the mean periosteal thickness was slightly greater in females, this finding was not significant (p=0.2875) (Figure 5). There was no significant variation in mean periosteal thickness between left and right femoral necks (p=0.5848).
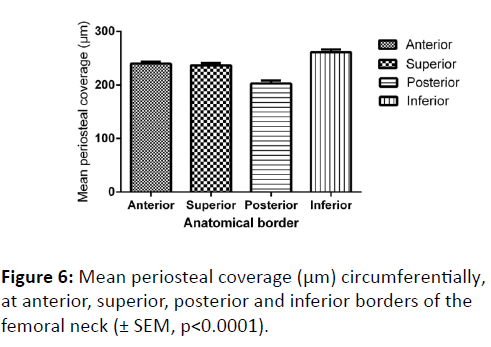
In terms of mean periosteal thickness, a comparison between the four quadrants (anterior, superior, posterior and inferior) found that there was a highly significant difference in thickness, with the posterior quadrant having the thinnest covering, and the inferior the greatest (p<0.0001) (Figure 6). Nevertheless, a comparison between males and females of periosteal thickness at each of the four quadrants found there to be no significant variation between the sexes.
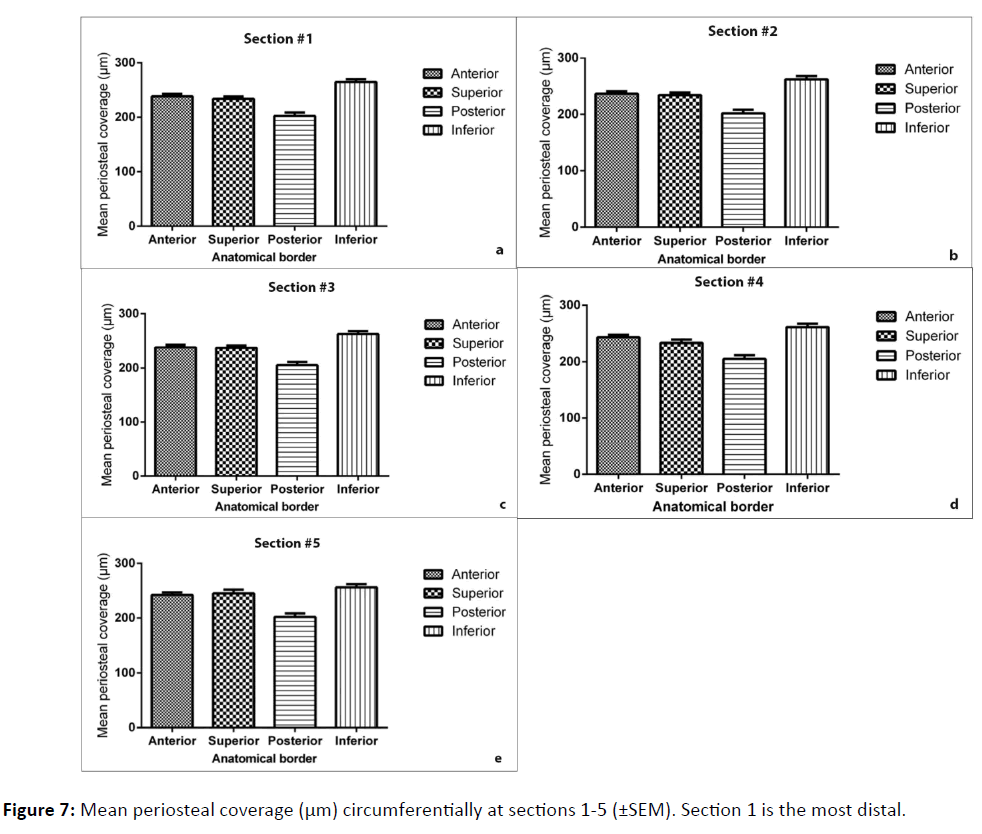
Finally, the coverage of periosteum was further analysed by comparing quadrant coverage on individual sections along the length of the femoral neck (Figure 7). The inferior border was found to be consistently the thickest region, and the posterior was the thinnest (p<0.0001). Variation between sequential segments along the length of the neck was found to be not significant (p=0.9594).
Discussion
The risk of osteoporotic fractures increases with old age [13]. Therefore, it might be expected that coverage of periosteum at the femoral neck would decline with age. Furthermore, since sex is another osteoporotic risk factor, it might be expected that females would possess thinner periosteal coverage than males, due to the effects of menopause and subsequent oestrogen deficiency on osteoblast proliferation [14]. Contrary to expectations, the data reported here suggest that neither of these suppositions is in fact borne out by the experimental evidence.
Further analysis carried out to examine the periosteal coverage circumferentially and along the length of the femoral neck confirms our hypothesis that periosteal coverage varies circumferentially. We found that the periosteum is thickest at the inferior border which may be a consequence of loadbearing during gait.
During normal gait, the subcapital and mid-femoral neck regions of the proximal femur withstand the greatest biomechanical forces. Compressive stress increases propensity of fracture whereas tensile stress is less of a risk at the femoral neck [13]. A study by Lotz et al. [15] found that compressive stress upon the femoral neck is greatest inferiorly when walking, with low tensile force existing superiorly.
Periosteal apposition is also thought to occur with age, as a compensatory mechanism to increased mechanical stress [16,17]. Since the femoral neck experiences high compressive stress inferiorly through a lifetime of walking, quiescent periosteal tissue may be induced to resume bone apposition [18]. This mechanism confers strength to the femoral neck under stress and may account for the results obtained in this study.
Mean periosteal coverage was found to be thinnest at the posterior border, which may have been due to the disruption of the posterior periosteum on the femoral neck seen particularly in Salter-Harris type II fractures of the femur in children [18]. It could also be argued that due to minimal compressive stress posteriorly while walking [15], the periosteum in this region is not stimulated to grow, thus remaining relatively thin throughout life.
Mean periosteal coverage against age of death showed positive correlation, contrary to our initial expectations. Power et al. [6] also reported age-related periosteal bone formation at the femoral neck, as a result of elevated alkaline phosphatase expression; periosteal apposition can also increase due to increasing mechanical stress upon the femoral neck with age, as suggested by a number of studies [16-21].
Limitations
The sample size of 9 cadavers was too small to detect differences of periosteal coverings between the sexes. Indeed, the difference recorded was so minimal that a power calculation suggested that over 800 cadavers would be necessary to detect a significant difference – clearly way beyond the scope of this study. The data for variation round the circumference, however, was highly significant. Also, although the relevant medical history for all the donors was sought, it would have been useful to have access to a full history of past medications.
Conclusion
Age and sex appear to have no influence on the periosteal coverage of the femoral neck. However, there are significant differences in coverage round the circumference, with the posterior quadrant being the most sparsely covered, and the inferior quadrant having the thickest covering, consistent with the theory that these regions are subject to the least and greatest compressive forces during gait, respectively. This distribution is consistent along the length of the femoral neck.
There is much potential for research regarding the periosteum, with possible techniques to minimise the incidence of femoral neck fractures in the elderly. Pharmacological interventions, such as those used to increase bone density, could be targeted to maintain and increase periosteum thickness in osteoporotic bones. Furthermore, the manipulation of chondrogenic cells may in future preserve the proliferative potential of the periosteum with age. Krebsbach et al. [22] successfully induced fibroblasts with bone morphogenetic protein-7 (BMP-7) to produce bone in vivo through gene therapy-directed osteogenesis. Similarly, it has been observed that fibroblasts, osteoblasts and progenitor cells of the periosteum can be successfully grown in vivo and in vitro [23], and potentially transplanted into periosteum at sites where it is thinning.
Acknowledgements
We gratefully acknowledge the deceased individuals involved in this study, without whom this work would not have been possible.
Funding
This study was supported by St George’s, University of London, and received no outside funding.
References
- Dwek JR (2010) The periosteum: what is it, where is it, and what mimics it in its absence? Skeletal Radiol 39: 319-323.
- Mackie EJ, Ahmed YA, Tatarczuch L, Chen KS, Mirams M (2008) “Endochondral ossification: how cartilage is converted into bone in the developing skeleton,” The International Journal of Biochemistry and Cell Biology40: 46-62.
- Balena R, Shih MS, Parfitt AM (1992) “Bone resorption and formation on the periosteal envelope of the ilium: a histomorphometric study in healthy women,” Journal of Bone and Mineral Research7: 1475-1482.
- O'Driscoll SW, Saris DB, Ito Y, Fitzimmons JS (2001) The chondrogenic potential of periosteum decreases with age. J Orthop Res 19: 95-103.
- Johnson ND, Wood BP, Noh KS, Jackman KV, Westesson PL, et al. (1989) MR imaging anatomy of the infant hip. AJR Am J Roentgenol 153: 127-133.
- Power J, Loveridge N, Rushton N, Parker M, Reeve J (2003) "Evidence for bone formation on the external "periosteal" surface of the femoral neck: a comparison of intracapsular hip fracture cases and controls," Osteoporosis International14: 141-145.
- Allen MR, Burr DB (2005) Human femoral neck has less cellular periosteum, and more mineralized periosteum, than femoral diaphyseal bone. Bone 36: 311-316.
- Orwoll ES (2003) Toward an expanded understanding of the role of the periosteum in skeletal health. J Bone Miner Res 18: 949-954.
- Shea JE, Vajda EG, Bloebaum RD (2001) Evidence of a hypermineralised calcified fibrocartilage on the human femoral neck and lesser trochanter. J Anat 198: 153-162.
- Albright F, Smith PH, Richardson AM (1941) "Postmenopausal osteoporosis," Journal of the American Medical Association116: 2465-2474.
- Seeman E (2007) The periosteum--a surface for all seasons. OsteoporosInt 18: 123-128.
- Walker DA, Linton AE, Schafer CT (1974) “Sudan Black B; a superior stain to Rose Bengal for distinguishing living from non-living foraminifera.” The Journal of Foraminiferal Research4: 205-215.
- de Bakker PM, Manske SL, Ebacher V, Oxland TR, Cripton PA, et al. (2009) "During sideways falls proximal femur fractures initiate in the superolateral cortex: evidence from high-speed video of simulated fractures," Journal of Biomechanics42: 1917-1925.
- Slootweg M, Ederveen A, Schot L, Schoonen W, Kloosterboer H (1992) "Oestrogen and progestogen synergistically stimulate human and rat osteoblast proliferation," Journal of Endocrinology133: R5-R8.
- Lotz J, Cheal E, Hayes W (1995) "Stress distributions within the proximal femur during gait and falls: implications for osteoporotic fracture," Osteoporosis International5: 252-261.
- Lazenby RA (1990) "Continuing periosteal apposition I: documentation, hypotheses, and interpretation," American Journal of Physical Anthropology82: 451-472.
- Beck TJ, Stone KL, Oreskovic TL, Hochberg MC, Nevitt MC, et al. (2001) "Effects of current and discontinued estrogen replacement therapy on hip structural geometry: the study of osteoporotic fractures," Journal of Bone and Mineral Research16 2103-2110.
- Matsumoto T, Nakayama K, Kodama Y, Fuse H, Nakamura T, et al. (1998) Effect of mechanical unloading and reloading on periosteal bone formation and gene expression in tail-suspended rapidly growing rats. Bone 22: 89S-93S.
- Kritsaneepaiboon S, Shah R, Murray MM, Kleinman PK (2009) "Posterior periosteal disruption in Salter-Harris Type II fractures of the distal femur: evidence for a hyperextension mechanism," American Journal of Roentgenology193: W540-W545.
- Krebsbach PH, Gu K, Franceschi RT, Rutherford RB (2000) Gene therapy-directed osteogenesis: BMP-7-transduced human fibroblasts form bone in vivo. Hum Gene Ther 11: 1201-1210.
- Arnsdorf EJ, Jones LM, Carter DR, Jacobs CR (2009) The periosteum as a cellular source for functional tissue engineering. Tissue Eng Part A 15: 2637-2642.
- Poole KE, Treece GM, Mayhew PM, Vaculík J, Dungl P, et al. (2012) Cortical thickness mapping to identify focal osteoporosis in patients with hip fracture. PLoS One 7: e38466.
- Evans SF, Chang H, Knothe-Tate ML (2013) "Elucidating multiscale periosteal mechanobiology: a key to unlocking the smart properties and regenerative capacity of the periosteum?" Tissue Engineering Part B: Reviews19: 147-159.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences