Normative Vertebral Hounsfield Unit Values and Correlation with Bone Mineral Density
Shaun P. Patel, John J. Lee, Garin G. Hecht, Sven A. Holcombe, Stewart C. Wang and James A. Goulet
DOI10.4172/2471-8416.100014
Shaun P. Patel1*, John J. Lee2, Garin G. Hecht3, Sven A. Holcombe4, Stewart C. Wang4 and James A. Goulet5
1Harvard Combined Orthopaedic Residency Program, Boston, MA, USA
2Department of Orthopaedic Surgery, Memorial Healthcare System, Hollywood, FL, USA
3Department of Orthopaedic Surgery, University of California - Davis, Sacramento, CA, USA
4Department of Surgery, University of Michigan, Ann Arbor, MI, USA
5Department of Orthopaedic Surgery, University of Michigan, Ann Arbor, MI, USA
- *Corresponding Author:
- Patel SP
Harvard Combined Orthopaedic Residency Program
55 Fruit Street, Boston
MA 02114, USA
Tel: 617-726-2942
E-mail: shaun.p.patel@gmail.com
Received date: March 25, 2016; Accepted date: April 07, 2016; Published date: April 11, 2016
Citation: Patel SP, et al. Normative Vertebral Hounsfield Unit Values and Correlation with Bone Mineral Density. J Clin Exp Orthop. 2016, 2:14 doi: 10.4172/2471-8416.100014
Copyright: © 2016, Patel SP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: We hypothesized that vertebral bone mineral density as measured in Hounsfield units (HU) by computed tomography (CT) can correlate with values obtained by dual x-ray absorptiometry (DXA). Our study aimed to report such a correlation along with the normative HU values within a large, heterogeneous population.
Methods: We identified 2,020 trauma patients with a CT scan encompassing any part of their thoracic or lumbar spine. Mean HU values within each vertebral body were reported with respect to vertebral level, sex, and age. Using linear regression analysis, our data were also compared to DXA results in patients having both CT and DXA scans.
Results: Analyses of CT scans yielded 28,083 unique vertebrae (18,489 male and 9,594 female) for which HU values were obtained. There were significant correlations between lumbar vertebral body HU and both DXA bone mineral density estimates (R2 = 0.50) and T-scores (p<0.001 and p<0.01, respectively). Using DXA T-scores as the reference standard, the sensitivity, specificity, and positive and negative predictive values of CT HU were 0.93, 0.75, 0.74 and 0.94, respectively.
Conclusion: CT can be used to measure vertebral bone mineral density and may aid in the diagnosis and management of osteoporosis.
Keywords
Bone mineral density; Osteoporosis; Osteopenia; Computed tomography; Hounsfield units; Dual xray absorptiometry; DXA
Background
The World Health Organization (WHO) defines osteoporosis as a bone mineral density more than 2.5 standard deviations below the young adult mean for a given population [1]. Patients with decreased bone mineral density have increased bone fragility and are at increased fracture risk [2]. As a consequence of our aging population, the public health burden of osteoporosis has reached epidemic proportions [3]. In 2004, 10 million Americans carried the diagnosis of osteoporosis, and the prevalence of osteopenia reached 34 million [4]. Every third postmenopausal woman and every fifth man older than 50 years is estimated to have osteoporosis [5]. Consequently, the incidence of osteoporotic fractures was greater than 2 million in 2005, including over 500,000 vertebral compression fractures [6]. The economic burden of osteoporotic fractures accrues $18 billion in direct medical costs annually [7], and this number is expected to grow by 150% by the year 2025 [6].
Bone densitometry is an important component of assessing fracture risk [8]. When compared to women at peak bone mass, results of densitometry testing have been shown to correlate to both fracture risk and efficacy of treatment [9]. Dual x-ray absorptiometry (DXA) is widely accepted to be the gold standard for measuring bone mineral density. Recently, the WHO’s Fracture Risk Assessment Tool (FRAX) has incorporated DXA T-scores into a more comprehensive fracture risk assessment [10]. However, DXA is an expensive test and is not without radiation. Accordingly, routine use of DXA screening in the presence of other significant indicators of osteoporosis has come into question [11].
Other methods of bone mineral density quantification include calcaneal ultrasound and quantitative computed tomography (qCT) [12]. A less expensive modality, peripheral bone ultrasound has been shown to correlate with fracture risk. Similar to DXA, though, it is a unique imaging study dedicated only to assessing bone mineral density. Conversely, qCT is not a stand-alone study and can be used to assess fracture risk in patients already undergoing such imaging [13]. To date, the majority of studies comparing qCT to DXA have reported lower accuracy and precision. However, this was likely due to the need for regular phantom calibration as a means to establish reference measures for CT scanners [14]. Since the advent of automatic exposure control in qCT, modern scanners now automatically account for a patient’s body habitus, leading to a more homogenous x-ray beam encountered by bone. This theoretically eliminates the need for the phantom calibration that was used in most qCT studies and allows a more accurate and precise bone mineral density measurement [15]. As such, Schreiber et al. [15] were able to show that modern clinical qCT scanners can report Hounsfield unit (HU) measurements that correlate well with bone mineral density of trabecular bone and DXA T-scores.
While the financial cost and radiation exposure of CT make it unreasonable to order solely for assessing bone mineral density [12], numerous patients at risk for osteoporosis undergo CT scans of the chest, abdomen, or pelvis for other clinical reasons [16]. The goal of our study is to report normative HU measurements per vertebral level of a large, heterogeneous population of otherwise healthy trauma patients. This normative set of data does not currently exist for a large cohort representing the general population. Additionally, no data exist on vertebral bone density outside the L1-L4 range reported by DXA, which could be useful for assessing fracture risk if the population norms are known. Our study’s data will help standardize CT HU measurements to DXA bone mineral density estimates in order to aid in the diagnosis and management of osteoporosis.
Methods
This study was approved by the [blinded] Institutional Review Board. We identified 2,020 trauma patients over the age of 19 years at the [blinded] between 2001 and 2011 who underwent a CT scan encompassing any part of their thoracic or lumbar spine. We did not exclude any of these patients in order to maintain a large, heterogeneous population representative of the general population. The CT scanners used during this study period were equipped with automatic exposure control (General Electric, Waukesha, WI).
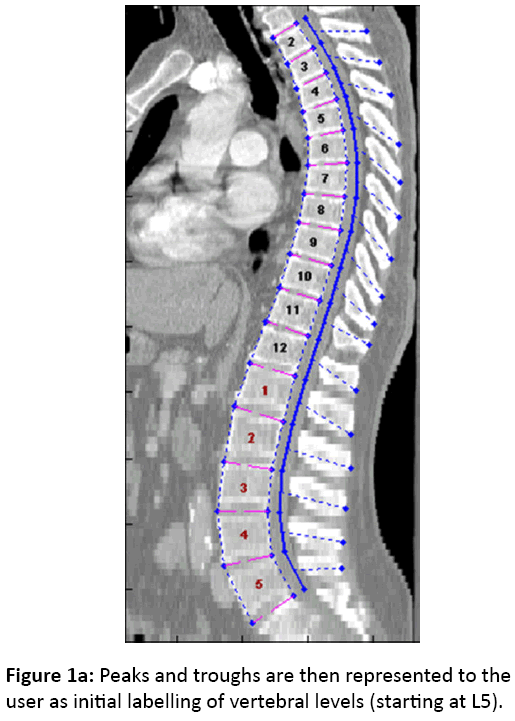
We developed an automatic processing script using MATLAB R2011a (Math Works, Natick, MA) that generated crosssectional slices of each vertebral body from which the average HU values were obtained. This involved the identification of the curve of the spine from the spinal canal, identifying and labelling individual vertebral levels and identifying the anterior and posterior vertebral bodies as points of reference (Figure 1a). Cross-sectional slices were then taken of each vertebra using these points of reference.
Identifying the spinal canal begins with an automated process based on the proximity of the spinal canal to any surrounding vertebral bone. A 3-dimensional distance map is calculated from bone (specified as 200 HU or higher). Candidate canal centers are identified as planar local maxima in the distance map that are closer than 20 mm from any surrounding bone. These candidate locations are connected via image ridges in the distance map volume, and an estimated 3-dimensional curve is fitted, approximating the spinal canal. A graphical interface is provided to the user who may adjust the final curve manually in the sagittal and coronal planes. Using interpolated slices perpendicular to the spinal canal, the axial rotation of the spine as a function of caudal level is specified as the angle of the axis of symmetry of slice pixels within 40 mm of the spinal canal center.
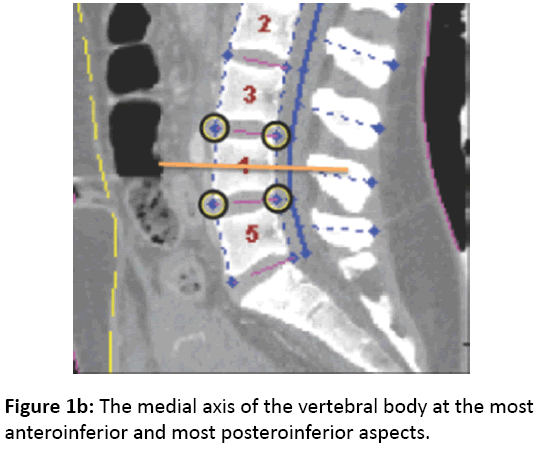
Next, a curved planar reformation (CPR) is created in the sagittal plane based on the 3-dimensional canal curve previously identified, aligned with the local axial twist. Traveling cranially along the CPR in 1 mm increments from the lowest spinal level imaged, density samples are taken between 5 and 30 mm anterior to the canal. Peaks in this signal correspond to vertebral body endplates, and hypodense troughs correspond to disc spaces. Peaks and troughs are then represented to the user as initial labelling of vertebral levels (starting at L5), and the user can manually correct any labelling errors (Figure 1a). At each specified vertebral level, two 3- dimensional points are recorded along the medial axis of the vertebral body at the most anteroinferior and most posteroinferior aspects (Figure 1b).
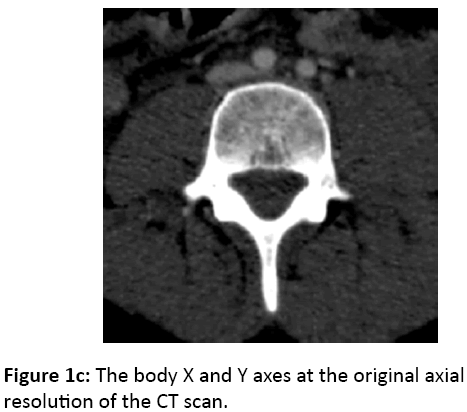
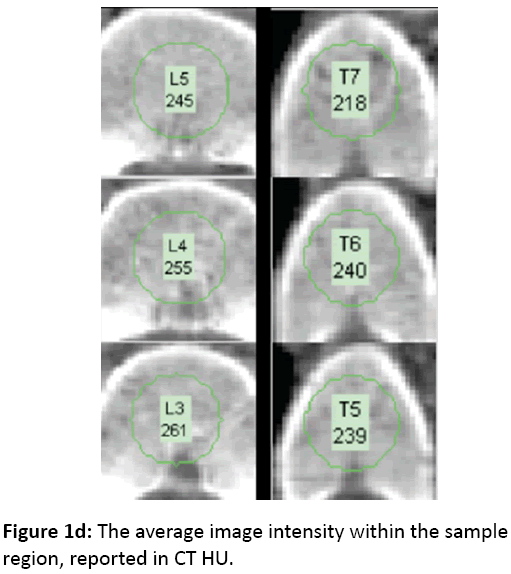
Vertebral HU measures are taken from a reconstructed cross-sectional slice as defined by the point selection process described above. These points define a coordinate system on the vertebral body with the origin located at the midpoint of the points and the Z axis representing the cranial-caudal axis of the spine. A new image slice is then created through the body X and Y axes at the original axial resolution of the CT scan, using cubic linear interpolation of the original volume (Figure 1c). A circular sample region of this slice is used, centered at the body origin and having a diameter equal to 75% of the average of distances posteroinferior-toanteroinferior, and posterosuperior-to-anterosuperior. The density of the vertebra is calculated as the average image intensity within the sample region, reported in CT HU (Figure 1d).
L1-L4 bone mineral densities from DXA scans were reported in two formats: automated reads from the scanners as well as radiology reads from clinical radiologists. These radiologists removed vertebra outliers from the final analysis to provide a more accurate report (e.g., removed vertebra with significant osteophytes or adjacent tissue calcification).
Mean HU are reported with respect to vertebral level, sex and age. Correlations between HU values and bone mineral density from DXA scans were calculated with use of the Pearson correlation coefficient. The relationship between Tscore groups (as defined by the WHO) and corresponding HU values was determined using one-way analysis of variance (Microsoft Excel, Redmond, WA). The sensitivity, specificity, and positive and negative predictive values for HU were calculated using the automated DXA report as the reference standard. Bone density was dichotomized as normal or abnormal on the basis of whether the HU value was less than the 95% confidence interval for subjects with normal T-scores ≥ -1.0.
Results
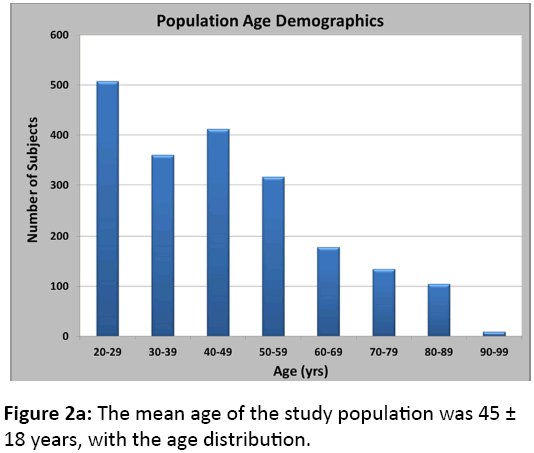
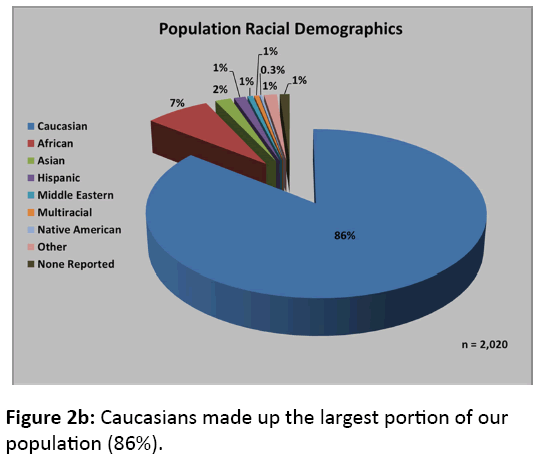
Of the 2,020 CT scans meeting our inclusion criteria, 1304 were from males (65%) and 716 were from females (35%). The mean age of the study population was 45 ± 18 years, with the age distribution shown in Figure 2a. Caucasians made up the largest portion of our population (86%), as demonstrated in Figure 2b. After processing the CT scans using the algorithms described earlier, we obtained data on 28,083 unique vertebrae (18,489 male and 9,594 female).
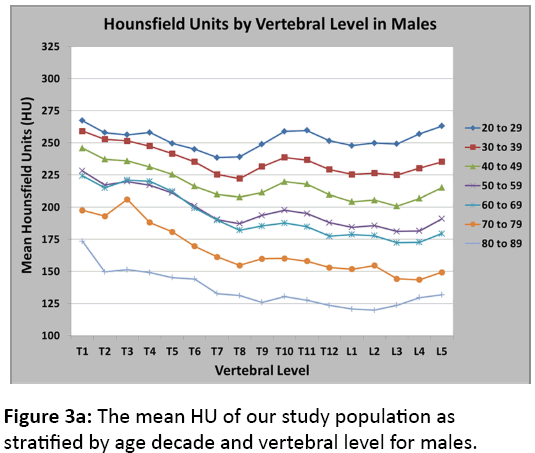
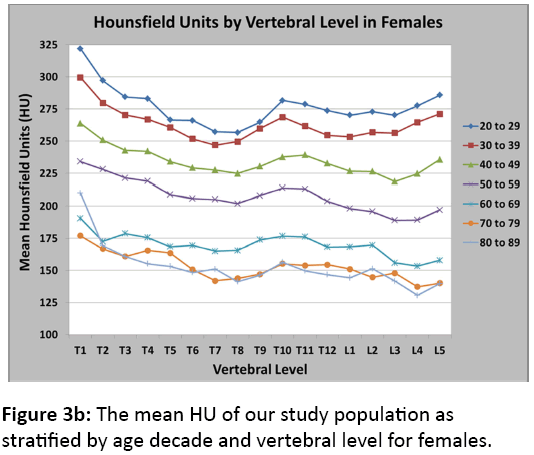
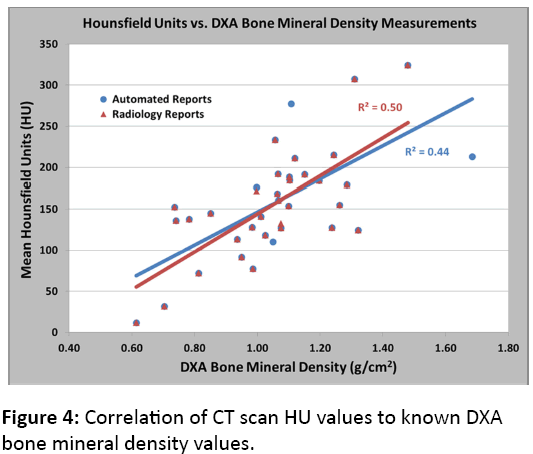
Figures 3a and 3b illustrate the mean HU of our study population as stratified by age decade and vertebral level for males and females, respectively. To validate our CT results and correlate them to DXA results, we identified a subset of postmenopausal female patients who underwent both a CT scan and a DXA scan within 90 days of each other (n=35). After linear regression analysis, we found a significant correlation between HU and bone mineral density as measured by DXA (Figure 4). This significant correlation was present when using both the DXA automated reports of L1-L4 (R2=0.44, p<0.001) and the DXA radiology reports (R2=0.50, p<0.01) where a radiologist reviewed the DXA scans and modified the automated reports using the methods noted above. Additionally, Table 1describes the relationship between HU values and T-scores produced by both DXA automated and radiologist-modified reports corresponding to normal, osteopenia, and osteoporotic bone mineral densities.
| Hounsfield Units | |||||
| DXA Automated Reports | DXA Radiology Reports | ||||
| Classification | T-Score | Mean ± SD | 95% CI | Mean ± SD | 95% CI |
| Normal | Greater than -1.0 | 195.7 ± 55.5 | 171.4 – 220.0 | 189.3 ± 58.9 | 159.5 – 219.1 |
| Osteopenia | Between -1.0 and -2.5 | 118.9 ± 29.1 | 98.7 – 139.0 | 139.4 ± 48.8 | 109.2 – 169.7 |
| Osteoporosis | Less than -2.5 | 97.9 ± 58.8 | 54.4 – 141.5 | 107.2 ± 60.4 | 65.3 – 149.0 |
Table 1: Mean and 95% confidence intervals of normal, osteopenia, and osteoporotic subjects.
Of those who had DXA and CT studies within 90 days of each other, each was determined to have either normal or abnormal bone density as determined by the lower limit of the normal 95% confidence interval from the DXA radiology reports (159.5 HU), using the T-score as the standard. The sensitivity, specificity, and positive and negative predictive values were 0.93, 0.75, 0.74 and 0.94, respectively, using the automated L1-L4 DXA scores as the reference and 0.77, 0.67, 0.72 and 0.71 using the radiologist’s DXA score report as the reference. Three DXA studies (8.6%, 3/35) were considered unreliable by a clinical radiologist secondary to significant sclerosis and/or osteophytes. The automated read differed from the radiology read 12.5% (4/32) of the time. When the unreliable DXA scans were also included, 20% (7/35) of the automated DXA reads were either considered unreliable or had differing reads from the radiologist. In the four cases in which the radiology read differed from the automated read, three of them were reported normal by the automated read but were considered osteopenia by the radiologist’s read. The single remaining study was considered osteopenia by the automated read and osteoporotic by the radiology read.
Discussion
To our knowledge, this study is the first to report normative Hounsfield units for the thoracic and lumbar spine for a large, heterogeneous population. This work establishes a reference database of Hounsfield units for each vertebral level using an automatic exposure control CT scan. Furthermore, we demonstrate an automated methodology that can quickly calculate bone mineral density using any CT scan that includes a portion of the vertebral spine. This methodology was tested and validated by our significant correlation of CT scan HU values to known DXA bone mineral density values (Figure 4). Moreover, our correlation was similar to previously reported results that utilized methodology not as automated as our own [15].
The ultimate goal of bone mineral densitometry is to better estimate fracture risk and subsequently guide treatment. DXA has become the standard of care in assessing patients at risk for osteoporotic fractures. However, DXA has numerous limitations, including its inability to discern between cortical and trabecular bone. While cortical and trabecular bone have both been shown to contribute to vertebral load sharing, trabecular bone bears the majority of the load throughout each vertebra [17]. Additionally, trabecular bone comprises the metabolically active portion of the bone most affected by pharmacotherapy for osteoporosis, such as bisphosphonates [18,19]. DXA results are also affected by bone size, because DXA uses planar techniques to estimate density [14,20]. Accordingly, larger bones have falsely elevated densities reported by DXA, which creates a gender difference trend on DXA that is not consistent with previously reported quantitative CT normative values [21]. These limitations of DXA support the investigation of other bone densitometry modalities and may also in part account for our DXA to CT correlation values of 0.44 and 0.50, which, although statistically significant, indicate only a moderate correlation.
In the United States, most insurers, including Medicare, cover initial DXA screenings in high-risk populations, such as women over the age of 65 years. In other countries, peripheral ultrasounds have been introduced as a dedicated imaging study that results in a cheaper investment than DXA scanners [22]. While CT is expensive and has unnecessarily high levels of radiation for a dedicated study to assess only bone mineral density, 30–40 million CT scans of the chest, abdomen, and pelvis are performed annually in the United States alone that could be used to assess bone mineral density at little or no additional cost [16]. Furthermore, CT scans are most often taken in older and frailer populations who are most at risk for osteoporosis and fragility fractures. Obtaining vertebral bone mineral density measurements from CT scans will allow more efficient diagnosis and treatment of osteopenia and osteoporosis, in addition to reducing the economic burden of singular dedicated densitometry studies such as DXA and peripheral ultrasound.
Our study is not without limitation. First, our population was comprised of trauma patients at the [blinded] who underwent CT scans between 2001 and 2011. This population best approximates the general population, but is still biased by being selected from a single center in [blinded], where the number of people with osteoporosis and osteopenia is estimated to rise at a rate below the national average over the next 10 years [4]. Next, our methods for averaging HU values in each vertebra take an average value of 75% of the area making up a perpendicular slice through a given vertebra. This does not fully account for the well-documented heterogeneity of trabecular bone [23]. However, our DXA to CT correlation was still very similar to those reported by Schreiber et al. [15], despite our differences in methodology for averaging HU values. Additionally, multiple iterations of our own methodology showed that taking a larger percentage of the perpendicular slice did not significantly change the HU values recorded. Furthermore, a number of CT scanners were used in our study and the uniformity of the automatic exposure control protocols may have varied. The calibration of these devices is unknown to us but was performed on a routine basis per the manufacturer’s recommendations.
The underlying comorbidities of patients were not included in our analyses, and many of these patients may have been on metabolic bone medications such as hormone replacement therapy, bisphosphonates, or teriparatide, potentially elevating the normative HU values for this group of patients. Separately analysing those who were on metabolic bone medications from those naïve may have elucidated the effects of these medications on vertebral HU. Furthermore, menopause could roughly be observed between the fifth and seventh decades of life, as seen in a proportionally larger loss of HU in figure 3b between these age groups. Between the fourth and fifth, fifth and sixth, and sixth and seventh decades of life, there was an average percentage loss of HU across all vertebral levels of 10.8%, 11.4%, and 18.2%, respectively.
When using the lower limit of the normal population HU from our correlation analysis with DXA bone mineral density (Table 1), 41% of those 50 years of age or older, 37% of those 60-69 years old, 60% of those 70-79 years old, and 67% of those 80-89 years old were considered osteopenia or osteoporotic based on their L1-L4 mean HU. These figures are lower than the estimate provided by the National Osteoporosis Foundation of 55% of those age 50 years or older having osteopenia or osteoporosis [4]. This may be due to the aforementioned regional bias in our patient population and our lower female to male ratio of 0.7:1, with fewer males having abnormal bone density. When stratified by sex, 45% (138/308) of females and 37% (166/443) of males 50 years or older were considered osteopenia or osteoporotic by HU.
The severe morbidity and mortality of osteoporotic fractures are often underappreciated, and the societal cost of these fractures is enormous [3,24]. While population studies have revealed that treating patients with osteopenia and osteoporosis can reduce fracture rate, there is still much to learn about fracture risk at different vertebral levels [7,9,10]. Our study identified varying bone mineral densities across different vertebral levels within not only similar age ranges but also within the same patient. Further studies are necessary to help identify particular vertebral levels at increased risk for fracture, which could create opportunities for prophylactic support systems and stabilization devices rather than sole reliance on metabolic therapy. This increased understanding of bone mineral density at different levels may also have implications for predicting hardware failure in instrumentation [25].
Fracture prevention is the ultimate goal of clinical bone densitometry. With this work, we introduce a novel methodology to ascertain bone mineral density using CT scans. Our resultant findings suggest that CT scans can be used as a supplemental tool for measuring bone mineral density, and may subsequently aid in the diagnosis and management of osteoporosis.
References
- World Health Organization (1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech Rep Ser. 843: 1-129.
- Prentice A (2004) Diet, nutrition and the prevention of osteoporosis. Public Health Nutr. 7: 227-243.
- Office of the Surgeon General (2004) Bone health and osteoporosis: a report of the Surgeon General. Rockville, MD: Office of the Surgeon General (US.
- National Osteoporosis Foundation (2002) America’s bone health: the state of osteoporosis and low bone mass in our nation. Washington, DC: National Osteoporosis Foundation.
- Olszynski WP, Davison KS, Adachi JD (2004) Osteoporosis in men: epidemiology, diagnosis, prevention, and treatment. ClinTher. 26: 15-28.
- Burge R, Dawson-Hughes B, Solomon DH (2007) Incidence and economic burden of osteoporosis-related fractures in the United States, 2005-2025. J Bone Miner Res. 22: 465-475.
- Dell RM, Greene D, Anderson D (2009) Osteoporosis disease management: What every orthopaedic surgeon should know. J Bone Joint Surg Am. 6: 79-86.
- NIH Consensus Development Panel (2001) Osteoporosis prevention, diagnosis, and therapy. JAMA 285: 785-795.
- Marshall D, Johnell O, Wedel H (1996) Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ 312: 1254-1259.
- Kanis JA, Johnell O, Oden A (2008) FRAX and the assessment of fracture probability in men and women from the UK. Osteoporos Int. 19: 385-397.
- Mueller D, Gandjour A (2009) Cost-effectiveness of using clinical risk factors with and without DXA for osteoporosis screening in postmenopausal women. Value Health 12: 1106-1117.
- Lewiecki EM, Borges JL (2006) Bone density testing in clinical practice. Arq Bras EndocrinolMetabol 50: 586-595.
- Lang TF, Keyak JH, Heitz MW (1997) volumetric quantitative computed tomography of the proximal femur: precision and relation to bone strength. Bone 21: 101-108.
- Guglielmi G, Lang TF (2002) Quantitative computed tomography. SeminMusculoskeletRadiol. 6: 219-227.
- Schreiber JJ, Anderson PA, Rosas HG (2011) Hounsfield units for assessing bone mineral density and strength: a tool for osteoporosis management. J Bone Joint Surg Am. 93: 1057-1063.
- Brenner DJ, Hall EJ (2007) Computed tomography--an increasing source of radiation exposure. N Engl J Med. 357: 2277-2284.
- Eswaran SK, Gupta A, Adams MF (2006) Cortical and trabecular load sharing in the human vertebral body. J Bone Miner Res. 21: 307-314.
- Black DM, Greenspan SL, Ensrud KE (2003) the effects of parathyroid hormone and alendronate alone or in combination in postmenopausal osteoporosis. N Engl J Med. 349: 1207-1215.
- Rubin MR, Cosman F, Lindsay R (2002) the anabolic effects of parathyroid hormone. Osteoporos Int. 13: 267-277.
- Kanis JA, McCloskey EV, Johansson H (2008) A reference standard for the description of osteoporosis. Bone 42: 467-475.
- Genant HK, Block JE, Steiger P (1987) Quantitative computed tomography in assessment of osteoporosis. SeminNucl Med. 17: 316-333.
- Magkos F, Manios Y, Babaroutsi E (2005) Quantitative ultrasound calcaneus measurements: normative data for the Greek population. Osteoporos Int. 16: 280-288.
- Keaveny TM, Hayes WC (1993) a 20-year perspective on the mechanical properties of trabecular bone. J Biomech Eng. 115: 534-542.
- National Osteoporosis Foundation (2009) Osteoporosis Annual Report. Washington, DC: National Osteoporosis Foundation.
- Turkyilmaz I, Ozan O, Yilmaz B (2008) Determination of bone quality of 372 implant recipient sites using Hounsfield unit from computerized tomography: a clinical study. Clin Implant Dent Relat Res. 10: 238-244.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences