O3DC: The Curiosity in Orthopaedics
Mohamed Mediouni, Amal Khoury
DOI10.4172/2471-8416.100010
Mohamed Mediouni1* and Amal Khoury2
12500 Boul. De l'Université, Sherbrooke, Quebec J1K 2R1, Canada
2Department of Orthopaedics, Hadassah Hebrew University Medical Center, Jerusalem, Israel
- *Corresponding Author:
- Mediouni M
2500 boul. de l'Université
Sherbrooke, Quebec J1K 2R1, Canada
E-mail: mohamed.mediouni@USherbrooke.ca
Received date: January 22, 2016; Accepted date: March 07, 2016; Published date: March 10, 2016
Citation: Mediouni M, et al. O3DC: The Curiosity in Orthopaedics. J Clin Exp Orthop. 2016, 2:10 doi: 10.4172/2471-8416.100010
Copyright: © 2016 Mediouni M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
To explore Mars, NASA sent a robot ''Curiosity'' which has many goals including Martian climate, geology, and the preparation for future human exploration. NASA has successfully overcome many challenges in terms of the complexity of the technology, Mars conditions and space problems. The mission of Curiosity has a powerful takehome message for the orthopaedic community: the design of a realistic training simulator is possible, which requires time, resources, and great effort. In this article, we detail orthopaedic 3D collection (O3DC). This paradigm was the result of discussion of the challenge in orthopaedic simulations discussed by many scientists.
Keywords
Orthopaedic simulation; Training; Fracture; Medical imaging
O3DC: Strategies
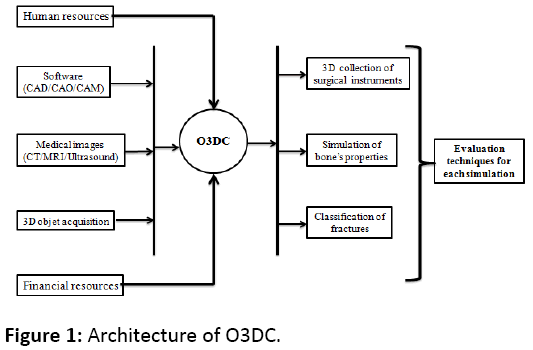
The great challenge of the orthopaedic innovation is to develop a transition strategy in a large scale, in order to transport the advanced achievements in the technology framework and the pilot projects for the benefit in research. Mediouni et al. [1] mentioned the trends and challenges in orthopaedic simulation. For that purpose, we need a universal common language that facilitates international exchanges. Orthopaedic 3D Collection (O3DC) is a platform that facilitates the design of the training simulator. The architecture of the platform, which has three principal aims is illustrated in figure 1:
• A 3D database of all surgical instruments (section 2).
• A good comprehension of biology can help to simulate the different levels of bone (section3).
• Using a 3D simulation, we can redefine a new classification of fractures (section 4).
Based on these three aims, the surgeons can evaluate the performance of the simulation, in which we are able to make a difference between expert and novice, otherwise between the experimented and inexperienced orthopaedist. O3DC is a multidisciplinary project that needs many researchers in different fields (orthopaedic, biology, mathematics, physics and computer science). Today, to ensure achievement of O3DC, we need a translational researcher [2] to identify new research discoveries in the biological field and clinical problems. He must ensure coordination between researchers in a transparent, organized and structured way.
3D Collection of Surgical Tools
Among the challenges to perform a simulator is the unavailability of 3D surgical instruments. In the literature, there are several 3D databases for industry such as Aim @ Shape [3], The Stanford 3D Scanning Repository [4], and 3D Warehouse [5]. Today, to achieve an efficient orthopaedic simulator, we need a specific database for surgical instruments which can be accessible online. 3D objects can be performed by a technology of acquisition such as a Kinect camera [6] and a 3D scanner or 3D modelling software [7]. The visual quality plays an important role in the efficiency of the simulation. The different stages of pre-processing are described by Mediouni [8] (Figure 2).
Simulation of Bone's Proprieties
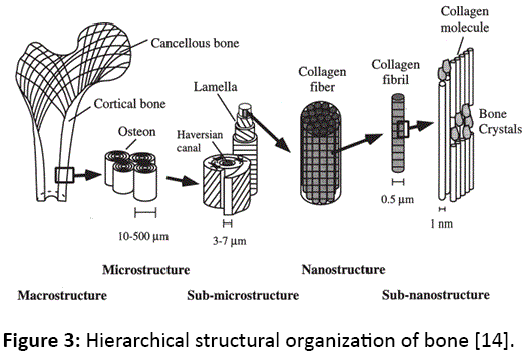
The bone has a homogeneous and complex material structure at many length scales. In order to understand mechanical properties, we must distinguish the different levels of hierarchical structure of the bone. Figure 3 shows four levels which are: (1) macrostructure, (2) microstructure, (3) submicrostructure, (4) nanostructure. The simulation of mechanical properties needs a model that explains the mechanism of interaction between different layers to improve surgeons' knowledge in materials and structural level. At macrostructure, the properties depend on the nature of the bone (cortical and cancellous). For example, the structural properties are important to determine the global stress analyses. At microstructure, osteoporosis is the subject of active research. The osteons properties were determined for various stress modes: tension [9], compression [10], shear [11] and flexion [12]. To complete the microstructural analysis of bone tissue, the evaluation of mechanical properties of cortical bone seems necessary, particularly as the number of studies on the human cortical bone is relatively low. At submicrostructure, the mechanical property is a major element in understanding bone anisotropy. On the scale of the nanostructure, the bone tissue can be considered as a composite material constituted of an organic matrix in which are inserted minerals. According to Fratzl et al. [13], hydroxyapatite crystals grow essentially in the intermolecular spaces of the collagen fibrils. The largest dimension of the crystals is aligned along the axis of the collagen fibrils. The shape, location and quantity of mineral extrafibrillaire remain unknown and subject to controversy despite the presence of crystals on the surface of the fibrils.
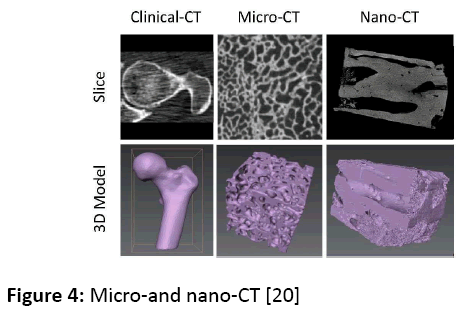
Using medical imaging becomes an important tool to simulate the bone architecture. Odgaard [15] discusses the usefulness of three-dimensional imaging to quantify trabecular architecture (Figure 4), which helps surgeons for studying the mechanical proprieties of cancellous bone, we need a compressive test. We can distinguish two major problems which are: (1) the absence of the standard model that can describe completely the mechanical proprieties, (2) the results provided are not of good quality for different reasons: temperature effects [16], storage [17], and viscoelasticity [18]. Parkinson et al. [19] have mentioned many features that we can distinguish from a 3D representation of cancellous bone: bone volume, density, trabecular thickness, and trabecular separation.
In addition, other features, such as degree of anisotropy, structural model index and connectivity density, can be calculated. With all the layers and the complexity of the characteristics, which include different details, the simulation of a bone remains a major challenge in orthopaedics.
Classification of Fractures
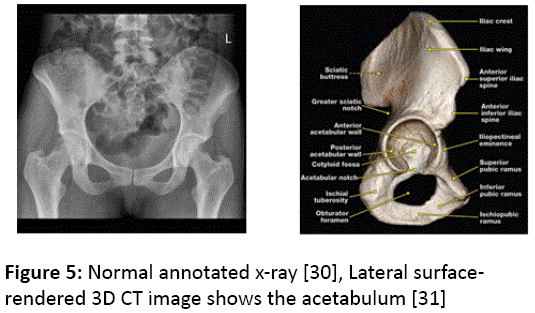
There are over 6.2 million bone fractures in the U.S. each year [21]. The recognition and classification of fractures become a necessity for an effective treatment. The Judet and Letournel system [22], which is a traditional system based on the X-ray, does not answer to complex fractures. Today, with the 3D simulations, radiologists must reclassify fractures. Matsushigue [23] explains 3D reconstruction images (CT) to improve the classification of the proximal extremity of the humerus. Using the alphanumeric system, the Arbeitsgemeinschaft für Osteosynthesefragen Association grouped the humerus fractures into 27 groups (Figure 5). 3D (CT) provides many advantages to identify the fracture and help surgeons make a decision for the treatment. Many studies of 3D imaging of acetabular fractures have been cited in the literature [24-29]. This fracture is tricky because of the complex spatial anatomy.
Evaluation of Simulation
In literature, there are some evaluation models of surgical simulations [32-36]. Unfortunately, there is no standard model to validate the assessment followed by orthopeadic community. Among the O3DC missions, is to provide an evaluation model for the gestures performed by surgeons to reduce each fracture (simple and complex). Generally, the validation is performed in the presence of a surgeon who discusses the effect of the simulation with his students [37]. Today, we can speak about the development of metric methods that can allow simulators to be autonomous. Sewell et al. [38] discuss the method of automated feedback in the context of a mastoidectomy simulator. The question arises: Is it possible to apply this model in orthopaedics?
Conclusion
This project is a call for orthopeadists to provide more research in 3D medical images to align with the complexity of fractures and diseases in the bone. The sentence mentioned by Dr. Pedowitz [39] can summarize our article:
“I think the changes we are going to see in resident education will be quite profound over the next decade or so.”
References
- Mediouni M, Volosnikov A (2015) The trends and challenges in orthopaedic simulation. Journal of Orthopaedics 12: 253-259.
- Woolf SH (2008) The Meaning of Translational Research and Why It Matters. AMA 299: 211-213.
- The shape repository. News: visionair.
- The Stanford 3D Scanning Repository.
- https://3dwarehouse.sketchup.com/?hl=en
- Zeng W (2012) Microsoft Kinect Sensor and Its Effect. IEEE Computer Society 4-10.
- Falkingham PL (2012) Acquisition of high resolution 3D models using free, open-source, photogrammetric software. Palaeontologia Electronica 15: 1.
- Mediouni M (2016) Orthopaedic 3D Collection: Mission and Impact. M J Ortho 1: 004.
- Ascenzi A, Bonucci E (1982) The tensile properties of single osteonic lamellae : Technical problems and preliminary results. Journal of Biomechanics 15: 29-37.
- Ascenzi A, Benvenuti A, Bonucci A (1967) The compressive properties of single osteons. Anatomy Recordings, 158: 377-392.
- Ascenzi A, Bonucci E (1967) The shearing properties of single osteons. Anatomy Recordings 158: 499-510.
- Ascenzi A, Baschieri P, Benvenuti A (1990) The bending properties of single osteons. Journal of Biomechanics 23: 763-771.
- Fratzl P, Gupta HS, Paschalis EP, Roschger P (2004) Structure and mechanical quality of the collagen-mineral nano-composite in bone. Journal of Materials Chemistry 14: 2115-2123.
- Rho JY, Kuhn-Spearing L, Peter Zioupos (1998) Mechanical properties and the hierarchical structure of bone. Medical Engineering & Physics 20: 92-102.
- Odgaard A (1997) Three-Dimensional Methods for Quantification of Cancellous Bone Architecture 20: 315-328.
- Brear K, Currey JD, Raines S, Smith KJ (1998) Density and temperature effects on some mechanical properties of cancellous bone. Eng Med 17: 163-167.
- Sedlin ED, Hirsch C (1966) Factors affecting the determination of the physical properties of femoral cortical bone. ActaOrthopScand 37: 29-48.
- Linde F (1994) Elastic and viscoelastic properties of trabecular bone by a compression testing approach. Danish Med Bull 41: 119-138.
- Parkinson IH, Forbes D, Sutton-Smith P, Fazzalari NL (2010) Model-Independent 3D Descriptors of Vertebral Cancellous Bone Architecture. Journal of Osteoporosis 1-6.
- Abel RL, Prime M, Jin A, Cobb JP, Bhattacharya R (2013) 3D Imaging Bone Quality: Bench to Bedside. Hard Tissue 2: 42.
- Velez M, Day T, He Y, Jung S Day DE (2012) Nano glass fibers and nano powders for medical applications. J NanomedicNanotechnol 3.
- Saterbak AM, Marsh JL, Turbett T, Brandser E (1995) Acetabular fractures classification of Letournel and Judet--a systematic approach. The Iowa Orthopaedic Journal 15: 184-196.
- Matsushigue T, Pagliaro Franco T, Pierami R , Jun Sugawara Tamaoki M, ArchettiNetto N, et al. (2014) Do computed tomography and its 3D reconstruction increase the reproducibility of classifications of fractures of the proximal extremity of the humerus? rev bras ortop 49: 174-177.
- Guy RL, Butler-Manuel PA, Holder P, Brueton RN (1992) The role of 3-D CT in the assessment of ac- etabular fractures. BrJRadiol 65: 384-389.
- Magid D, Fishman EK, Ney DR, Kuhlman JE, Frantz KM, et al. (1992) Acetabular and pelvic fractures in the pediatric patient: value of two- and three-dimensional imaging. J PediatrOrthop 12: 621-625.
- Martinez CR, DiPasquale TG, Helfet DL, Graham AW, Sanders RW, et al. (1992) Evaluation of acetabular fractures with two- and three-dimensional CT. RadioGraphics 12: 227-242.
- Scott WW, Fishman EK, Magid D (1987) Acetabular fractures: optimal imaging. Radiology 165: 537-539.
- Scott WW, Magid D, Fishman EK, Riley LH Jr, Brooker AF Jr, et al. (1987) 3-D evaluation of ac- etabular trauma. ContempOrthop 15: 17-24.
- White MS (1991) Three-dimensional computed tomography in the assessment of fractures of the acetabulum. Injury 22: 13-19.
- Pelvis. Bony pelvis: radiopaedia.
- Scheinfeld MH, Dym AA, Spektor M, Avery LL, Joshua Dym R, et al. (2015) Acetabular Fractures: What Radiologists Should Know and How 3D CT Can Aid Classification. Radiographics 35: 555–577.
- Shaharan S, Neary P (2014) Evaluation of surgical training in the era of simulation. World J GastrointestEndosc 6: 436-447.
- Ogata M, Dohi Y, Yamada T, Kubota Y (2012) Implementation and Evaluation of Hyperelastic Model for Surgical Simulator and Navigation. Engineering in Medicine and Biology Society (EMBC), 2012 Annual International Conference of the IEEE.
- Sigounas VY, Callas PW, Nicholas C, Julie E Adams, Daniel J Bertges, et al. (2012) Evaluation of Simulation-Based Training Model on Vascular Anastomotic Skills for Surgical Residents. Society for Simulation in Healthcare 7: 334-338.
- SangildSørensen T, Franz Greil G, Kromann Hansen O, Mosegaard J (2006) Surgical simulation – a new tool to evaluate surgical incisions in congenital heart disease? Interactive CardioVascular and Thoracic Surgery 5: 536–539.
- Elbashti ME, Hattori M, Sumita YI, Taniguchi H (2015) Evaluation of articulation simulation system using artificial maxillectomy models. Journal of Oral Rehabilitation.
- Mediouni M (2015)The What, Where and Why of Orthopaedic Simulation.Int J Surg Res Pract 2:3.
- Sewell C, Morris D, Blevins NH, Agrawal S (2007) Validating Metrics for a Mastoidectomy Simulator. Stud Health Technol Inform 125: 421-426.
- McKee J (2013) “Rebooting” Orthopaedic Resident Education. American Academy of Orthopaedic Surgeons.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences